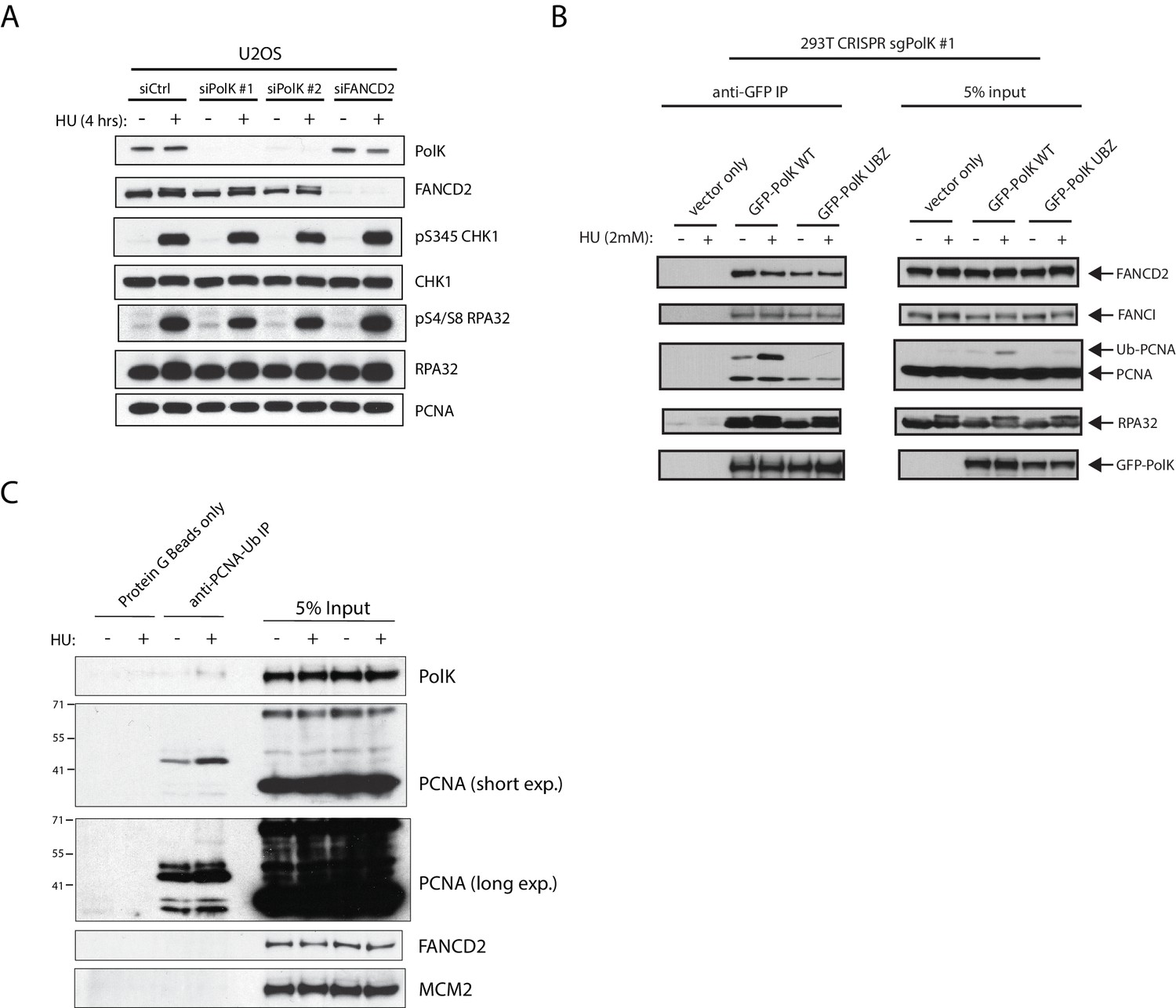
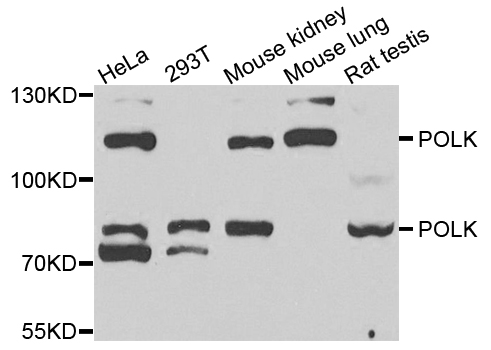
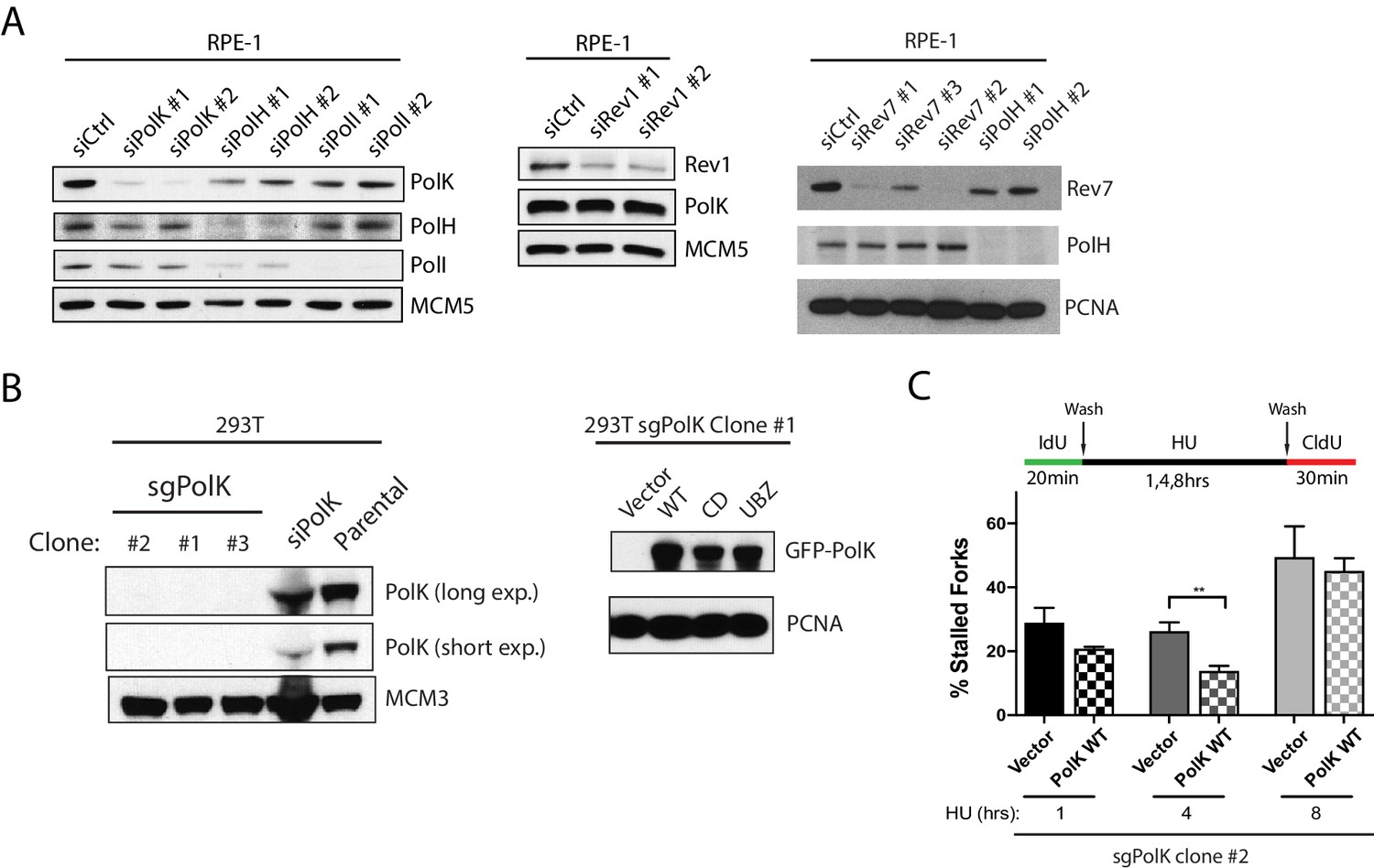
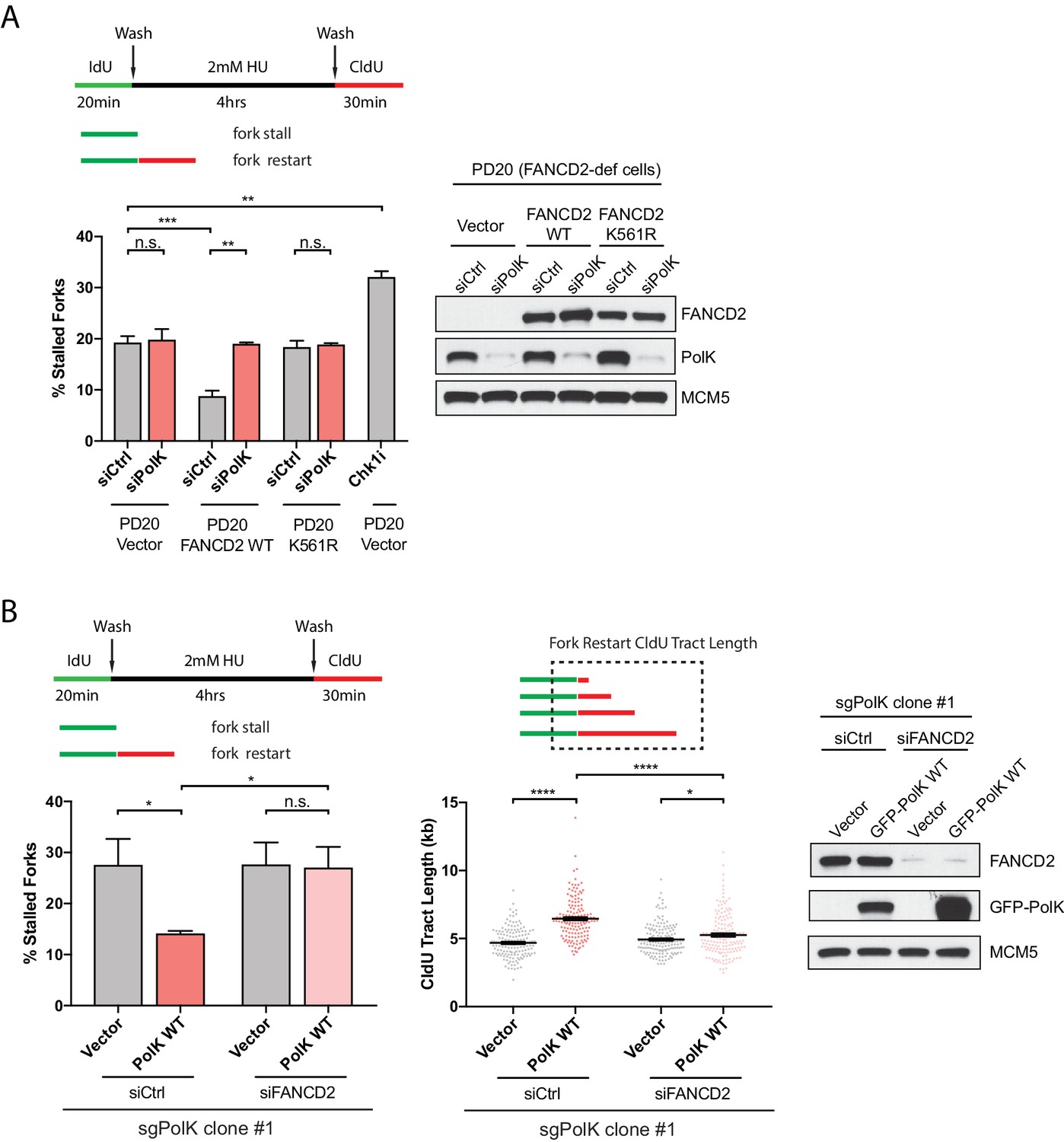
A catalytic-independent function of human DNA polymerase Kappa controls the pool of the Checkpoint Kinase 1 | bioRxiv

Phosphorylation Alters the Properties of Pol η: Implications for Translesion Synthesis - ScienceDirect

Mechanism of Replicative DNA Polymerase Delta Pausing and a Potential Role for DNA Polymerase Kappa in Common Fragile Site Replication - ScienceDirect
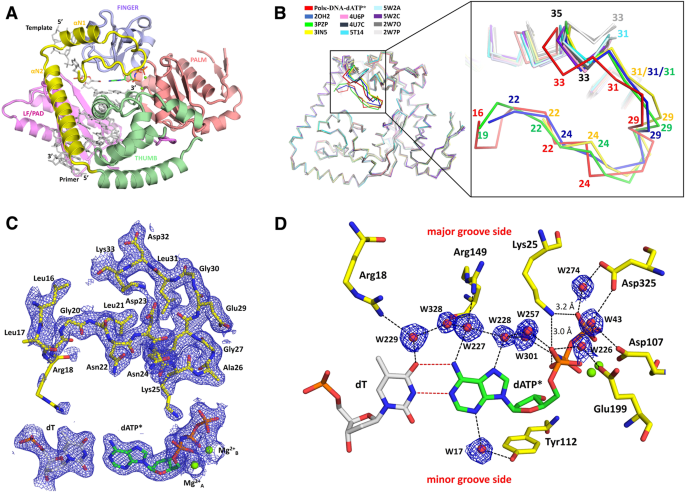
2.0 Å resolution crystal structure of human polκ reveals a new catalytic function of N-clasp in DNA replication | Scientific Reports

Translesion Synthesis of 2′-Deoxyguanosine Lesions by Eukaryotic DNA Polymerases | Chemical Research in Toxicology

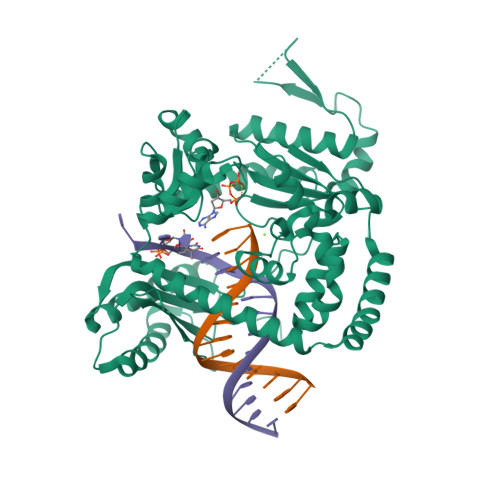
RCSB PDB - 6BS1: Crystal Structure of Human DNA polymerase kappa in complex with DNA containing the major cisplatin lesion

In vivo evidence that DNA polymerase kappa is responsible for error-free bypass across DNA cross-links induced by mitomycin C - ScienceDirect





![PDF] Mammalian DNA Polymerase Kappa Activity and Specificity | Semantic Scholar PDF] Mammalian DNA Polymerase Kappa Activity and Specificity | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b8e3a90ed2d17229033cb68c855f8761b2d83146/2-Figure1-1.png)